DATE
September 15, 2025
CATEGORY
Blog
SHARE

Introduction: Why This Approval is a Game-Changer
Tolebrutinib’s recent approval in the UAE marks a pivotal moment for the neurology therapeutics landscape in the Middle East and North Africa (MENA) region. As an oral Bruton’s tyrosine kinase (BTK) inhibitor approved for non-relapsing secondary progressive multiple sclerosis (SPMS), this development is not just a regulatory milestone—it signals a transformative shift in how the region is embracing innovation.
Tolebrutinib, developed by Sanofi, represents a major advancement in multiple sclerosis (MS) treatment as an oral, highly selective, and brain-penetrant Bruton’s tyrosine kinase (BTK) inhibitor. This approval will not only boost the BTK inhibitor market in the MENA region but also propel the BTK inhibitors market growth in the leading markets such as the US, EU4, the UK, and Japan.
Unlike conventional MS therapies that primarily act on the peripheral immune system, Tolebrutinib penetrates the central nervous system (CNS), targeting B cells and microglia—two key drivers of neuroinflammation and progression in MS. This unique mechanism addresses both inflammatory relapses and progressive disease components, making it particularly promising for patients with secondary progressive MS (SPMS) and primary progressive MS (PPMS). Tolebrutinib has demonstrated significant reductions in MRI lesions and shows potential for slowing disability progression, with a manageable safety profile. Its oral formulation enhances patient adherence compared to injectable therapies, while its dual action on adaptive and innate immunity positions it as a transformative therapy in the MS space.
The UAE’s approval of Tolebrutinib signals a strong regional commitment to early adoption of innovative treatments, providing Sanofi a first-mover advantage in the MENA market and setting the stage for other neurology players to leverage this momentum for accelerated access, strategic partnerships, and real-world evidence generation.
The UAE, with its proactive healthcare strategies, has positioned itself as a key gateway for pharmaceutical and biotechnology companies entering MENA. With multiple sclerosis prevalence rising across Gulf Cooperation Council (GCC) countries and an increasing government commitment to advanced neurological care, this approval sets the stage for new opportunities in neuroimmunology and CNS disorders.
The UAE Regulatory Landscape: Fast-Track for Innovation
The UAE’s Emirates Drug Establishment (EDE) has significantly modernized its regulatory processes over the past five years to position the country as a regional life sciences hub. The recent approval of Tolebrutinib exemplifies this shift toward agility and innovation.
Historically, regulatory timelines in the region could take 12 to 18 months, creating barriers for companies aiming to bring cutting-edge therapies quickly to patients. Today, fast-track approval pathways allow innovative treatments addressing high unmet needs to reach patients in as little as six months; a dramatic improvement in speed-to-market.
For pharma players, regulatory acceleration equals competitive advantage. The ability to secure earlier market entry means greater access to first-mover benefits, enhanced brand visibility, and deeper stakeholder engagement.
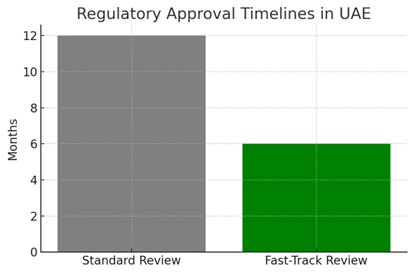
[ Visual 1: Regulatory Timelines Comparison Chart – Standard vs Fast-Track in UAE]
Why This Matters for Neurology Market Entrants
The approval of Tolebrutinib underscores two key dynamics: market potential and competitive landscape evolution.
Market Potential: Rising Burden of MS
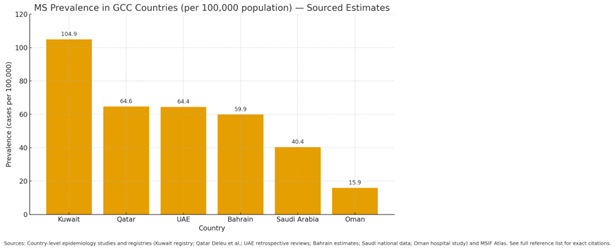
GCC countries, particularly the UAE and Saudi Arabia, have seen a steady rise in MS cases, attributed to better diagnostic capabilities, improved awareness, and demographic changes. Current estimates indicate MS prevalence ranging between 55 and 80 per 100,000 population across the GCC, with DelveInsight estimating roughly 6,000 prevalent cases in 2024 in the UAE alone, creating a sizable patient base for advanced therapies.
[ Visual 2: MS Prevalence in GCC Countries Chart]
Neurology: A High-Growth Therapeutic Area
The MENA neurology drug market is expected to experience unprecedented growth. An aging population, lifestyle changes, the launch of emerging therapies, and significant investment in healthcare infrastructure fuel this growth.
Tolebrutinib’s approval signals that regulatory and payer systems are now more receptive to breakthrough therapies, particularly in CNS and neuroimmunology.
Competitive Advantage for Early Movers
Sanofi, with Tolebrutinib, has taken a first-mover position in progressive MS therapy in the UAE, setting pricing benchmarks and access models that others will follow. For companies developing BTK inhibitors, S1P receptor modulators, or innovative monoclonal antibodies, this development is a strong indicator that the regulatory door is open for advanced neuro therapies.
Challenges to Watch Out For
While the opportunity is significant, market entrants should not underestimate the complexities of the MENA region. Key challenges include:
- Pricing Pressure: Innovative therapies often face pushback from payers and procurement bodies, particularly in tender-based markets like Saudi Arabia.
- Payer Dynamics: The UAE follows an insurance-driven model, while Saudi Arabia relies heavily on government tenders. Strategies must adapt to both.
- Market Access Hurdles: Health Technology Assessment (HTA) adoption is gaining traction in the region, requiring robust pharmacoeconomic evidence.
- Awareness & Diagnosis Gaps: Despite improvements, early diagnosis remains a challenge, limiting timely intervention.
Strategic Considerations for New Entrants
To succeed in this evolving market, companies need a strategic playbook that addresses regulatory, commercial, and patient-centric dimensions:
1. Regulatory Alignment
- Leverage fast-track approval pathways for therapies addressing high unmet needs.
- Maintain early engagement with local regulatory authorities to align submission strategies.
2. Market Access & Value Demonstration
- Develop strong value dossiers tailored to regional payer priorities.
- Prepare budget impact models to demonstrate long-term cost-effectiveness.
- Explore risk-sharing agreements to mitigate payer uncertainty.
3. Local Partnerships
- Collaborate with distributors, specialty clinics, and digital health platforms for localized execution.
- Identify Key Opinion Leaders (KOLs) to champion new therapies within healthcare ecosystems.
4. Patient Engagement Programs
- Implement patient support programs (PSPs) to improve adherence and outcomes.
- Drive education campaigns to bridge knowledge gaps among patients and caregivers.
Broader MENA Trends Neurology Companies Should Leverage
The Tolebrutinib approval reflects a broader set of trends reshaping the healthcare landscape in MENA:
- Digital Health Integration: Tele-neurology and remote monitoring solutions are gaining traction, driven by post-pandemic digital adoption.
- Precision Medicine Push: National genome projects in the UAE and Saudi Arabia aim to integrate personalized care approaches for chronic diseases, including neurology.
- Government Vision 2030 Strategies: Both the UAE and Saudi Arabia have prioritized chronic disease management, including neurological disorders, in their healthcare transformation roadmaps.
Turning Approvals into Commercial Success
While the regulatory door is open, market entry is not merely about securing approval—it’s about building sustainable access and adoption strategies. Companies that align early with regulators, design evidence-based access plans, and invest in patient engagement will capture long-term value.
From regulatory readiness assessments to pricing strategy development, a strategy-driven approach can help pharmaceutical companies translate scientific innovation into commercial success in MENA.
A Signal of What’s Next
Tolebrutinib’s approval in the UAE is more than a single drug launch rather it’s a bellwether for the future of neurology in MENA. It demonstrates regulatory agility, growing payer openness to innovation, and rising patient demand for advanced therapies.
For neurology players, the message is clear:
Act now. Align regulatory strategies. Build robust market access frameworks. Invest in patient engagement.
The next three years will define the leaders in MENA’s neurology market.








