DATE
November 7, 2024
CATEGORY
Blog
SHARE

Stem cell therapy is a groundbreaking field in regenerative medicine, using stem cells to repair or replace damaged tissues and organs. With the potential to treat a wide range of conditions—including degenerative diseases, injuries, and specific cancers—stem cells offer a promising therapeutic pathway. Due to their ability to differentiate into various cell types, stem cells are currently being evaluated in numerous clinical trials around the world.
Several types of stem cells are used in regenerative medicine, each with unique properties and applications:
- Embryonic Stem Cells (ESCs): Derived from early-stage embryos, these cells are pluripotent and can develop into any cell type.
- Adult Stem Cells: Found in adult tissues, these cells are more limited in differentiation but are useful in therapies for certain tissue repairs.
- Induced Pluripotent Stem Cells (iPSCs): Generated by reprogramming adult cells, iPSCs offer pluripotency similar to ESCs without the ethical concerns.
In recent years, the MENA region has made significant strides in stem cell research, led by substantial investments in R&D, particularly in the UAE. Dubai, for instance, has established the first regional Stem Cells Bank, positioning the UAE as a leader in stem cell research and a future hub for stem cell tourism. Initiatives such as the MENA Stem Cells Forum and the Dubai Stem Cell Congress bring together international and regional experts to exchange knowledge, fostering a collaborative environment aimed at advancing stem cell therapies in the region.
The regulatory landscape in the MENA region is evolving to support ethical and safe applications of stem cell therapies. The UAE leads in establishing specific regulations for clinical stem cell use, ensuring ethical practices in line with international standards. However, broader regional coordination is needed to harmonize guidelines across countries, facilitating cross-border research and treatments. Such harmonization would enhance research collaboration and support the development of a cohesive regulatory framework across the region.
Stem cell therapies are increasingly being adopted across the MENA region to treat various medical conditions, including diabetes, cardiovascular diseases, and neurological disorders. Efforts are focused not only on pioneering new therapies but also on making existing treatments more accessible to the local population. Notably, mesenchymal stem cells have shown promise in treating COVID-19, driving further interest in stem cell research.
While progress has been substantial, challenges remain:
- Regulatory Harmonization: Despite the UAE’s progress in regulatory development, a cohesive regional framework is necessary for standardizing ethical practices in stem cell research and therapy.
- Public Awareness and Acceptance: Varying levels of public awareness and acceptance of stem cell therapies highlight the need for education and outreach to build trust and understanding among the population.
- Research Infrastructure and Clinical Trials: Expanding clinical trials and research infrastructure is essential to fully leverage stem cell therapies for a broader range of diseases.
Stem cell therapy offers immense potential for transforming healthcare, particularly in the MENA region, where initiatives and investments are paving the way for future advancements. By addressing the challenges of regulatory harmonization, public awareness, and infrastructure development, the region can enhance its role as a global leader in stem cell therapies. With continued collaboration and commitment, stem cell therapy could significantly improve the health outcomes and quality of life for populations across the MENA region (Bahari et al., 2023).
List of major key players operating in MENA region
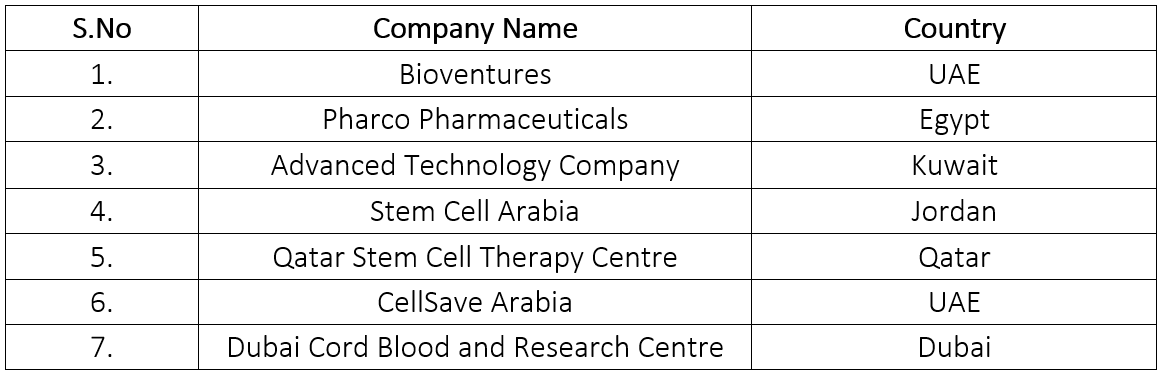
Progress in Stem Cell Therapy
- Establishment of various research centers and institutes: The progress in stem cell therapy has been facilitated by the development of advanced technologies for isolating, expanding, and manipulating stem cells for therapeutic purposes. Regional governments, particularly in the United Arab Emirates, Saudi Arabia, and Qatar, are dedicating significant funds to develop robust infrastructure for research and development in life sciences, including stem cell therapy. These investments have led to the establishment of specialized research centers or departments dedicated to stem cell research and therapy within hospitals. Many hospitals in the MENA regions now offer a wide range of stem cell-based treatments, attracting patients seeking advanced regenerative medicine options(Al-Shamsi et al., 2023).
List of various research centers in MENA regions
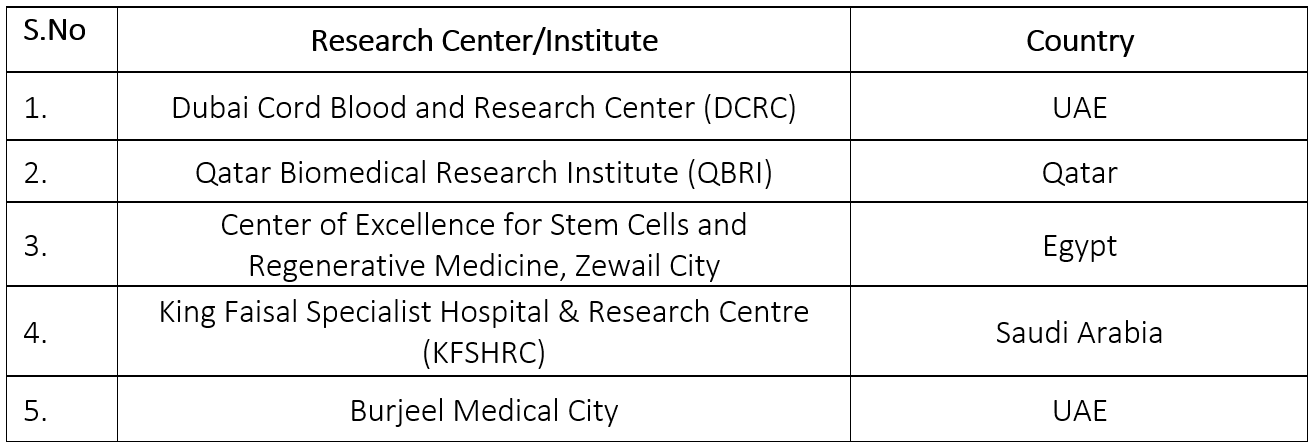
- Dubai Cord Blood and Research Center (DCRC), UAE: focus on stem cell research, banking, and therapeutic applications. DCRC plays a crucial role in advancing stem cell therapies and promoting public awareness about the importance of stem cell banking. It is a leading institution in the UAE, offering services such as cord blood banking and conducting clinical research(Dubai Health – Dubai Health, n.d.).
- Qatar Biomedical Research Institute (QBRI), Qatar: focus on stem cell therapy, diabetes research, and personalized medicine. QBRI, under Hamad Bin Khalifa University, is at the forefront of stem cell research in Qatar, with a strong emphasis on translating research into clinical applications, particularly in the field of diabetes treatment(QBRI | Hamad Bin Khalifa University, n.d.).
- Center of Excellence for Stem Cells and Regenerative Medicine, Zewail City, Egypt: focus on stem cell research, regenerative medicine, and biomedical sciences. The significance of this center is that it is a pivotal research hub in Egypt, dedicated to advancing stem cell science and its applications in treating diseases. It is also instrumental in fostering collaboration between researchers and clinicians(Center of Excellence for Stem Cells and Regenerative Medicine (CESC), n.d.).
- King Faisal Specialist Hospital & Research Centre (KFSHRC), Saudi Arabia: focus on stem cell transplantation, hematology, and regenerative medicine. KFSHRC is one of the leading medical research institutions in the region, with a dedicated department for stem cell transplantation. The center conducts cutting-edge research and offers advanced therapies for conditions such as blood disorders(KFSHRC, n.d.).
- Burjeel Medical City, UAE: focus on regenerative medicine and stem cell therapy and it is a key player in the MENA region’s stem cell therapy landscape, with ongoing research and clinical trials aimed at harnessing stem cells for treating various medical conditions, including cancer and degenerative diseases(Burjeel Medical City, n.d.).
These institutions are not only advancing scientific research but also playing a critical role in translating these advancements into clinical therapies, thereby improving patient outcomes in the MENA region.
- Government Support, Funding and Investment: Governments across the MENA region have increasingly supported stem cell research through strategic funding and investment. Although stem cell therapy has the potential to revolutionize healthcare in Saudi Arabia and globally, several critical challenges remain to be addressed. To mitigate public concerns about the safety and regulation of these therapies, as well as to improve understanding of ethical treatments and clinical trials, there is a need for enhanced education, communication, and transparency efforts. By fostering collaboration among government bodies, healthcare professionals, and research institutions, it is possible to build a more informed and trustworthy community, ensuring that patients have access to reliable and safe stem cell treatments. Some key developments includes the UAE government has made substantial investments in stem cell research and therapy, establishing specialized centers and departments within hospitals. The Abu Dhabi Stem Cell Center, for example, is a leading institution conducting collaborative research with international partners to develop innovative stem cell treatments. The Saudi government, through the National Guard Health Affairs Division, developed the Saudi Stem Cell Donor Registry (Aboalola & Ramadan, 2024). The country is also investing in public awareness and educational initiatives to minimize the lack of understanding about approved stem cell treatments. The Qatar Foundation has been actively supporting stem cell research through its Qatar Biomedical Research Institute (QBRI). QBRI focuses on translational research, including the development of stem cell-based therapies for various diseases.
- Clinical trials and ongoing Treatments: The MENA region has been increasingly active in conducting clinical trials for stem cell therapies, addressing a range of medical conditions. These trials focus on evaluating the safety, efficacy, and potential of stem cell treatments for chronic and complex diseases. Several countries in the region, including the UAE, Saudi Arabia, and Egypt, are leading these efforts, with numerous trials in progress. Many of these clinical trials are supported through collaborations with international research institutes. These partnerships help bring advanced technologies and expertise to the region, enhancing the quality and scope of the trials. Countries in the MENA region often partner with institutions in Europe and North America to conduct these studies, ensuring that the trials meet international standards and contribute to global scientific knowledge’s.
List of major institutions involved in stem cell research and clinical trials in the MENA region along with their specific areas of focus.
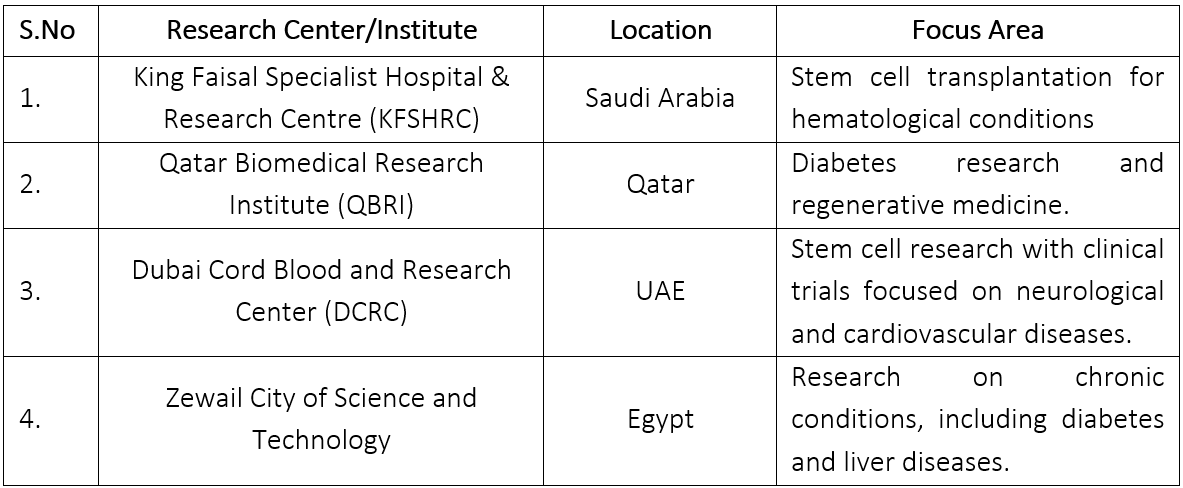
- International collaborations and Partnerships: International collaborations and partnerships have become increasingly vital to the advancement of stem cell therapy and biomedical research in the MENA region. These collaborations are essential for fostering innovation, enhancing research quality, and ensuring that the region stays at the forefront of scientific developments. MENA countries, particularly Saudi Arabia, the UAE, and Qatar, have established partnerships with renowned institutions worldwide. For example, the King Faisal Specialist Hospital & Research Centre (KFSHRC) in Saudi Arabia collaborates with prominent institutions such as the Mayo Clinic and Johns Hopkins Medicine. These partnerships facilitate the exchange of expertise, enabling local researchers and clinicians to gain exposure to advanced research methodologies and treatment protocols. The collaboration often includes joint training programs, workshops, and symposiums aimed at enhancing the skills of researchers and healthcare professionals in the MENA region(Mayo Clinic and King Faisal Specialist Hospital & Research Centre in Saudi Arabia Collaboration, 2018).
International collaborations often involve the transfer of advanced technologies to research centers in the MENA region. This includes sophisticated equipment for stem cell isolation, processing, and genetic engineering. For example, collaborations with European and American institutions have enabled MENA research centers to acquire cutting-edge tools and technologies essential for conducting high-quality stem cell research. MENA researchers and institutions frequently participate in global conferences and symposiums, where they present their findings and engage with the international scientific community. Events such as the MENA Stem Cells Forum provide a platform for networking and establishing new collaborations, allowing MENA researchers to stay updated on the latest developments in the field(4th MENA Stem Cells Forum , n.d.).
List of major companies, their work and technology involved in stem cell research and clinical development in the MENA region.
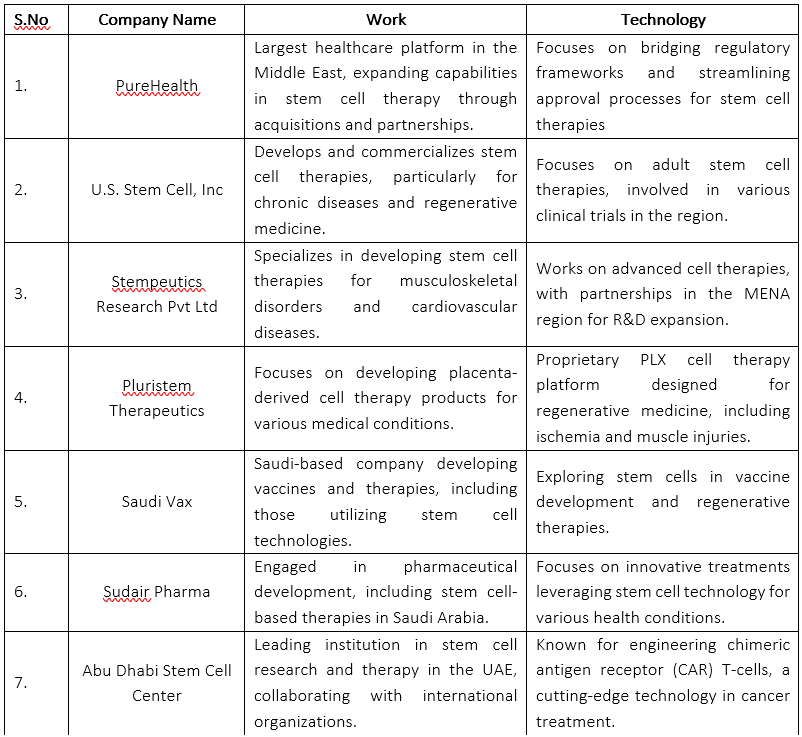
Pharmaceutical companies are playing a significant role in advancing stem cell research and therapy in the Middle East Pharmaceutical companies from the U.S., Europe, and Asia are collaborating with local research institutions in the Middle East to develop novel stem cell-based therapies tailored to the regional population. For example, PureHealth, the largest healthcare platform in the Middle East, completed the acquisition of Circle Health Group in the UK in 2024. This move is expected to enable PureHealth to bridge regulatory frameworks and streamline approval processes for stem cell therapies in the region(PureHealth Acquisition | Business Wire, 2024).
Challenges in Stem Cell Therapy
- Regulatory and Ethical Challenges: One of the main obstacles is the lack of a unified regulatory framework throughout the MENA region. Each country has its own regulations, which can differ widely. This inconsistency complicates the ability of companies and research institutions to navigate the regulatory environment, resulting in delays in the approval and commercialization of stem cell therapies. Additionally, stem cell research, especially that involving embryonic stem cells, raises ethical concerns that are particularly sensitive in the MENA region due to cultural and religious factors. Many countries impose strict regulations on the use of embryonic stem cells, which can restrict the breadth of research and development. In certain MENA nations, the regulatory landscape is complicated and not fully established, which can impede the advancement of clinical trials and the rollout of new therapies. This situation is often exacerbated by the absence of clear guidelines regarding the use of stem cells in medical treatments, creating uncertainty for researchers and clinicians(Dajani, 2014).
- Infrastructure and resource limitations: Limited access to advanced technologies remains a significant issue in the MENA region, despite some areas having advanced research capabilities. Access to essential technologies for stem cell research, such as cell culture, genetic engineering, and bio-preservation, is still restricted in many locations. Additionally, the infrastructure necessary to support high-level stem cell research and therapy is underdeveloped in various parts of the region. This includes the lack of specialized laboratories, biobanks, and facilities for conducting clinical trials, which can impede research progress and restrict the ability to perform large-scale clinical studies. Quality control is another critical challenge, as ensuring the consistency and quality of stem cell products is complicated by the absence of standardized protocols and facilities. Variability in the quality of these products can negatively impact treatment outcomes and undermine the credibility of stem cell therapies in the region(Enosawa, 2022).
- Financial challenges: Conducting stem cell research and therapy is financially demanding, necessitating considerable investment in research and development, clinical trials, and infrastructure. In the MENA region, funding from both government and private sectors is often inadequate, which restricts the extent of research and delays the introduction of new therapies(Hoang et al., 2022).
Although some governments in the region have begun to allocate funds for stem cell research, overall financial support remains low. The significant expenses associated with establishing and maintaining stem cell research facilities, coupled with limited funding, hinder researchers’ ability to carry out thorough studies. Investors in the MENA region generally exhibit a cautious approach, particularly regarding new and untested technologies like stem cell therapy. This hesitance to invest in high-risk research areas stifles industry growth and restricts the development of innovative therapies(Lo & Parham, 2009).
In conclusion, stem cell therapy in the MENA region is rapidly advancing, driven by significant investments in research and development, particularly in the UAE, Saudi Arabia, and Qatar. The establishment of specialized research centers, such as the Dubai Cord Blood and Research Center (DCRC) and the Qatar Biomedical Research Institute (QBRI), underscores the region’s commitment to pioneering stem cell therapies. Government support, coupled with international collaborations, has been instrumental in enhancing the region’s capabilities in stem cell research and clinical trials.
However, challenges remain, including the need for a unified regulatory framework, ethical considerations, and infrastructural and financial limitations. Addressing these challenges is crucial for the continued growth and success of stem cell therapy in the region, with the potential to revolutionize treatment options for a variety of medical conditions. The MENA region’s efforts in advancing stem cell therapies are poised to position it as a leader in regenerative medicine, although further coordination and investment are necessary to fully realize this potential.








